Specialized iNANO Lecture: Live-Cell Imaging at the Nanoscale
Dr. Emil B. Kromann, The Bewersdorf Laboratory, CT, USA
Info about event
Time
Location
iNANO meeting room 1590-213
Dr. Emil B. Kromann, The Bewersdorf Laboratory,
Dept. of Cell Biology, Yale School of Medicine, CT, USA.
Dept. of Biomedical Engineering, Yale Graduate School of Arts and Sciences, CT, USA
Live-Cell Imaging at the Nanoscale
Super-resolution imaging techniques have enabled researchers to take a closer look at sub-cellular structures and dynamics. I will briefly review two of these imaging techniques: Fluorescence PhotoActivation Localization Microscopy (FPALM aka. PALM/STORM), and Stimulated Emission Depletion (STED) microscopy. I will present recent advancements made in the Bewersdorf Lab, emphasizing my own work with live-cell STED microscopy:
Using stimulated emission depletion (STED) microscopy, we can image distinctly labeled structures with 20-30 nm resolution in fixed cells. Despite being built upon confocal microscopy (a powerful technique for observing live-cell dynamics) STED microscopy is rarely used to image living specimens. One of the most dominant reasons has been the lack of STED-compatible, photostable dye-pairs for intracellular labeling.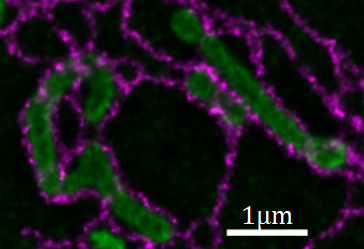
Figure 1: STED image (deconvolved) of mitochondria (green) and endoplasmic reticulum (magenta) in a living cell.
I will present STED instrumentation and a novel labeling strategy specifically geared towards two-color live-cell STED imaging of intracellular targets. In my instrument, I apply a spatial light modulator for beam shaping and aberration correction, FPGA based laser control, gated detection and a 16 kHz resonant scanning mirror, which reduces pixel dwell time and photobleaching. In combination with a novel dye pair my instrument enables continuous (up to 90 frames) two-color STED imaging of intracellular targets in living cells, with acquisition of one two-color STED image every two seconds (10 x 10 µm field of view, <50 nm resolution). See: Bottanelli, Kromann et al., Nature Communications, 2016.
Host: Associate professor Victoria Birkedal, iNANO & Dept. of Chemistry, Aarhus University
