Our research over the last years has covered different aspects of organic synthesis, mainly focusing on transition metal catalysis involving small gaseous building blocks, leading to the development of highly efficient methods for introducing isotope labels into pharmaceuticals. Furthermore, we have since 2015 been part of the Danish Nation Research Foundation Center of Excellence: Carbon Dioxide Activation Center (CADIAC), where the ultimate goal is to utilize carbon dioxide in the synthesis of valuable compounds, either via direct CO2 incorporation or CO2 reduction. And in 2022, we became part of the newly established Novo Nordisk Foundation CO2 Research Center (CORC), which deals with CO2 conversion in various aspects. Finally, we have recently moved into the field of polymer deconstruction, where we employ metal catalysis for the decomposition of plastic. Potentially, we would like to investigate the use of waste plastics and polymers for carbon capture.
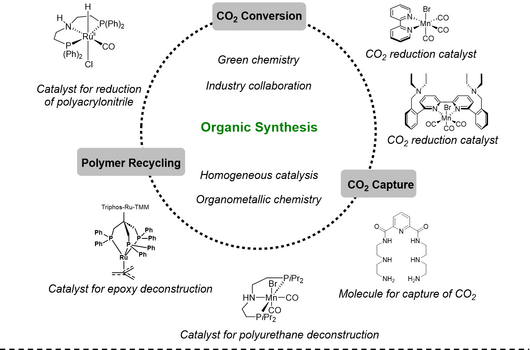
For more detalis about our ongoing research, have a look at our individual projects under funding, here you will be able to link directly to the projects we are a part of! Otherwise, browse through our publication list. If you would like to hear more about our ongoing research and/or reserach group in general, do not hesitate to contact us.