New publication in Synlett!
We have just published our work on a nickel-catalyzed carbonylative coupling of alkylzinc reagents and α-bromo-α,α-diflouroacetamides. Congratulations to all authors!
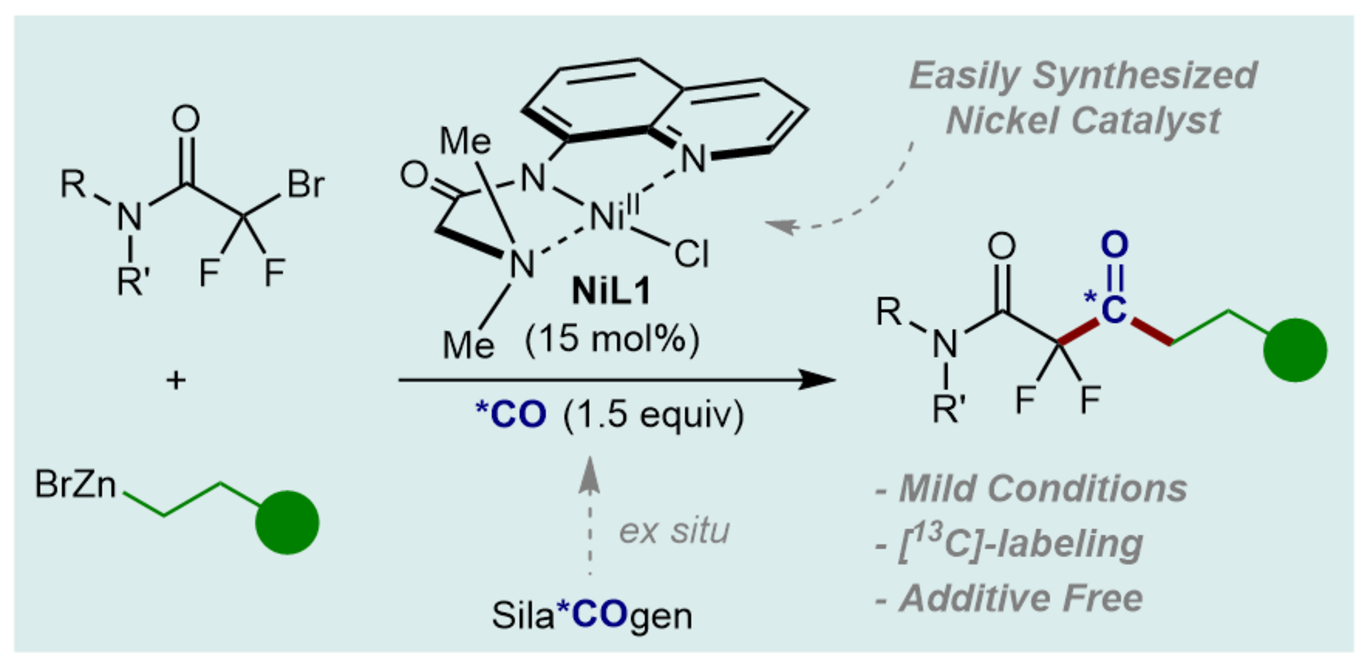
Abstract: Herein, we report a nickel catalyzed carbonylative cross coupling of alkyl zinc reagents and α,α-difluorobromoaceamides to obtain α,α,-difluoro-α-ketoamides in moderate to good yields. The reaction is catalyzed by a bench stable nickel(II) pincer complex, in contrast to other reports based on palladium catalysts. The carbonylative reaction is performed in a two-chamber system (COware®), in which carbon monoxide (CO) is generated ex situ from the solid precursor, SilaCOgen, and consumed in the adjacent chamber. The reaction operates at low temperatures using near stoichiometric amounts of CO, and isotopically labeled products are effortlessly accessed as demonstrated using 13C-labeled SilaCOgen.
https://www.thieme-connect.com/products/ejournals/abstract/10.1055/a-2036-4809