The ‘Laboratory for Cell Mimicry’ aims to employ bottom up assemble synthetic entities to assembled live-like units that could integrate with their natural counter parts. We work across the length scale, from synthesizing organic compounds as enzyme mimics over the assembly of catalytic nanoparticles as artificial organelles to the engineering of artificial cells and their integration with mammalian cells into bionic tissue.
Artificial organelles are nanosized reactors with intracellular activity, often encapsulated catalysis. In contrast to traditional pharmaceutical, artificial organelles are envisioned to provide a sustained solution for a chronic condition.
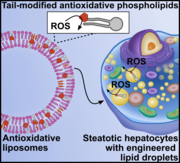
Intracellular reactive oxygen species (ROS) in steatotic cells can cause oxidative stress and damage. To address this, engineered phospholipids were delivered to intracellular lipid droplets in steatotic hepatic cells using the cells' lipid transport mechanisms. Tail-labeled fluorescent lipids formed into liposomes were shown to be transported to lipid droplets in steatotic HepG2 and HHL-5 cells. An antioxidant, a EUK salen–manganese derivative with superoxide dismutase and catalase activity, was covalently attached to a phospholipid tail and formulated as liposomes. Steatotic HepG2 and HHL-5 cells treated with these antioxidant liposomes had lower intracellular ROS levels compared to untreated controls and non-covalently formulated antioxidants. This study demonstrates a new strategy to equip native organelles in mammalian cells with engineered enzyme activity.
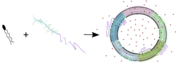
Amphiphilic block copolymers and lipids form hybrid vesicles (HVs), alternatives to liposomes and polymersomes. These copolymers, featuring hydrophobic poly(sitostryl methacrylate) or its copolymers with butyl methacrylate and hydrophilic poly(carboxyethyl acrylate), assemble with soybean L-α-phosphatidylcholine (soyPC) into small HVs. This assembly is confirmed by electron microscopy and small-angle X-ray scattering. Fluorescence resonance energy transfer and Laurdan fluorescence indicate a hybrid membrane structure similar to liposomes but with transmembrane asymmetry. Giant HVs with homogeneous copolymer and soyPC distribution, created via electroformation, show slower lateral diffusion and higher permeability compared to soyPC and sitosterol vesicles. This study advances the understanding of hybrid membranes combining natural and synthetic components.
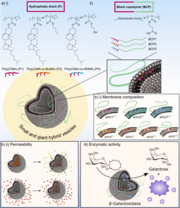
Hybrid vesicles (HVs), formed by combining phospholipids and amphiphilic block copolymers (BCPs), offer a contemporary alternative to liposomes and polymersomes. By altering the chemical composition of the hydrophobic block in the BCPs, specifically using statistical copolymers of cholesteryl methacrylate and either butyl methacrylate (BuMA) or 2-hydroxyethyl methacrylate (HEMA), variations in HV membrane properties were achieved. Membranes with HEMA-containing BCPs exhibited higher permeability, while those with BuMA provided an optimal environment for efficient association with β-galactosidase, demonstrating the significance of the hydrophobic block in controlling HV morphologies and properties.
Artificial cells are larger micron-sized assemblies that aim at structurally and functionally support mammalian cells and tissue.
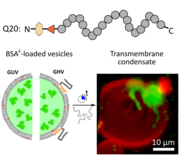
Biomolecular condensates and cellular membranes jointly regulate intracellular organization and membrane repair. Here, we use a minimal synthetic system to study how membrane composition and curvature control condensate–membrane interactions. Exposure of lipid or polymer–lipid hybrid giant vesicles containing bovine serum albumin to a resilin-inspired intrinsically disordered protein (IDP) undergoing liquid-liquid phase separation induced in situ formation of transmembrane condensates. This cargo-triggered condensation links encapsulated protein crowding to condensate nucleation and insertion. Condensate morphology depended on vesicle size and membrane elasticity: smaller vesicles and stiffer hybrid membranes favored transmembrane condensation, while larger or softer membranes promoted membrane wetting and deformation. Condensates also reorganized lipids locally and promoted leaflet coupling without disrupting membrane integrity or cargo retention. These findings provide mechanistic insight into membrane repair processes using a minimal model system.
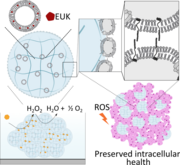
Integrating artificial and mammalian cells into semi-synthetic aggregates remains a challenge in synthetic biology. This study demonstrates the fabrication of cell membrane vesicles (CMVs) from HepG2 cells to coat alginate microgels, creating camouflaged artificial cells (ACs). These ACs facilitate the formation of synthetic and semi-synthetic aggregates. Predator-defendant and liver-like synthetic aggregates show promising potential, while camouflaged ACs enhance integration with HepG2 cells. Encapsulating a ROS-scavenging artificial enzyme in ACs protects HepG2 cells from oxidative stress, improving viability, proliferation, and mitochondrial health. This work advances the fusion of mammalian cells and ACs, with the latter serving as support units.

Protein-mediated division in synthetic systems remains a major challenge in bottom-up synthetic biology. Here, the reconstitution of the Drs2p-Cdc50p lipid flippase in hybrid vesicles enables lipid flipping and induces transmembrane asymmetry, with vesicle constriction and division depending on the hydrophobic block chemistry. This work represents a step toward mimicking cell division in synthetic assemblies and developing bottom-up self-replicating units.
Self-propelled particles attract a great deal of attention due to the auspicious range of application nanobots can be used for. In a biomedical context, self-propelled swimmers hold promise to autonomously navigate to a desired location in an attempt to counteract cell/tissue defects either by releasing drugs or performing surgical tasks. Self-propelled particles attract a great deal of attention due to the auspicious range of application nanobots can be used for. In a biomedical context, self-propelled swimmers hold promise to autonomously navigate to a desired location in an attempt to counteract cell/tissue defects either by releasing drugs or performing surgical tasks.
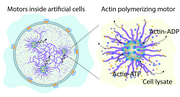
This work demonstrates the creation of artificial cells capable of forming cytoskeleton-like networks through actin polymerization driven by nanomotors. By mimicking the actin-based propulsion mechanism of Listeria monocytogenes, these hybrid vesicle-based systems integrate active matter and bottom-up synthetic biology to achieve life-like movement and structural organization.
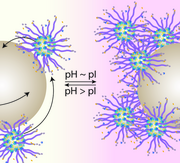
Nano- and micromotors are active colloids capable of self-propulsion beyond Brownian motion, but synthetic propulsion strategies are often limited by challenging reaction conditions. Inspired by natural biopolymerization-driven locomotion, we investigated silica motors functionalized with the actin-recruiting protein ActA from Listeria monocytogenes using homogeneous, Janus-type, and polymer-brush immobilization strategies. Janus-functionalized motors showed up to a 5-fold increase in propulsion over Brownian motion and exhibited pH-dependent interactions with larger tracer particles, demonstrating how biologically inspired design can enable unique synthetic micromotors.
The field of micro/nanorobots has progressed from science fiction to reality, with promising applications in biomedicine, environmental remediation, and industry, yet practical deployment remains limited. This review highlights key challenges such as propulsion, collective behavior, material design, scaling, and regulatory frameworks, hindering their widespread use. A future roadmap emphasizes interdisciplinary collaboration to overcome technical, ethical, and commercial barriers, positioning micro/nanorobots as vital tools for global challenges and improved quality of life.
Soft actuators are flexible, deformable devices designed to mimic the motion and adaptability of organisms or muscles. They are made from soft, responsive materials, enabling them to bend, stretch, and contract in response to external stimuli. These actuators are used in applications requiring gentle and adaptive movements, such as soft robotics, wearable devices, and biomedical devices.
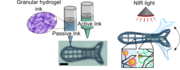
Stimuli-responsive soft actuators are central to soft robotics, enabling programmable shape changes in response to environmental cues. Here, we report a light-responsive fish-shaped actuator fabricated by 3D printing granular alginate-based hydrogel inks infiltrated with PNIPAM and gold nanostars for thermoresponsive and photothermal actuation. The resulting anisotropic structures showed reversible NIR-induced bending, with a fish-shaped actuator exhibiting reproducible actuation over nine cycles and a stabilized bending angle of ~37°, demonstrating the potential of modular granular inks for remotely controlled soft robotic systems.

Hydrogel-based actuators, responsive to external stimuli, are promising for applications like soft robotics. We use 3D printing to create layered rectangles with gelatin methacryloyl (GelMA) and GelMA-poly(N-isopropylacrylamide) (PNIPAM) inks, evaluating their bending capabilities in a buffer. Gold nanostars act as photothermal elements, enabling repeated bending of a 3D-printed fish tail under near-infrared light, demonstrating the potential of combining 3D printing, responsive hydrogels, and photothermal elements for soft robotics.